InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the “AI-Powered Real-Worl Evidence (RWE) Solutions Market”-, By Component(Software/Platforms, Services), By Data Source(EHR/EMR Data, Claims Data, Genomics, Wearables/IoT, Patient-Reported Data, Imaging), By Therapeutic Area(Oncology, Neurology, Cardiology, Rare Diseases, Immunology), By End-User (Pharma/Biotech, Payers/PBMs, Hospitals/IDNs, Regulators, CROs), By Deployment Model (Cloud-Based, On-Premise, Hybrid), By Technology (NLP, Computer Vision, Federated Learning, Graph ML), By Application (Drug Development, Regulatory Submissions, Market Access, Precision Medicine), and Global Forecasts, 2025-2034 And Segment Revenue and Forecast To 2035.”
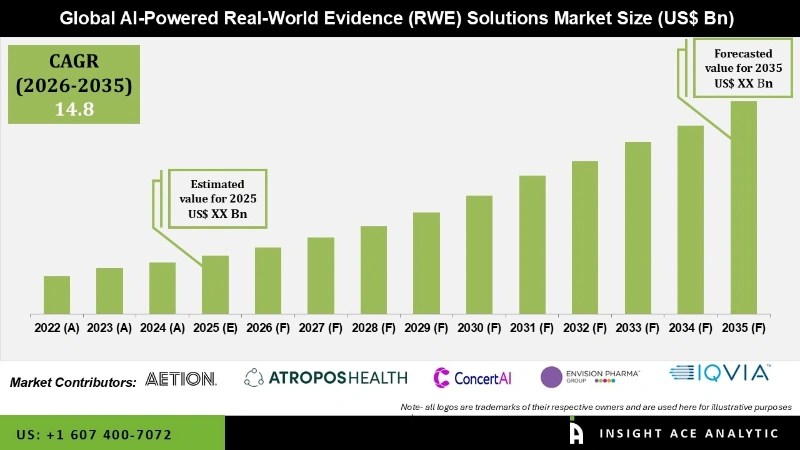
Global AI-Powered Real-World Evidence (RWE) Solutions Market Size is predicted to reach grow at a 14.8 % CAGR during the forecast period for 2026 to 2035.
Get Free Access to Demo Report, Excel Pivot and ToC: https://www.insightaceanalytic.com/request-sample/3135
Real-World Evidence (RWE) is generated from Real-World Data (RWD), which comprises health-related information collected outside the scope of conventional randomized controlled trials. Key sources of RWD include electronic health records (EHRs), insurance claims databases, patient registries, wearable technologies, and patient-reported outcomes. The effective utilization of these diverse data sources enables improved patient outcomes, more efficient drug development processes, and informed decision-making across the healthcare ecosystem. The integration of artificial intelligence (AI) into RWE generation is significantly transforming clinical trial design and execution within the pharmaceutical industry.
Pharmaceutical organizations are increasingly leveraging real-world patient data to gain deeper insights into treatment patterns, disease progression, and population variability, which are often not fully captured in traditional clinical trials. AI technologies are being employed to enhance study design through the optimization of inclusion and exclusion criteria, simulation of trial scenarios, and identification of suitable patient cohorts.
This approach improves patient recruitment efficiency, minimizes the need for protocol modifications, and reduces overall clinical trial costs. For instance, leading pharmaceutical companies have applied AI-driven analytics to large-scale historical clinical datasets, enabling improved patient stratification, accelerated study design, and enhanced competitiveness in early-stage development.
Regulatory bodies have also introduced frameworks to support the incorporation of RWE in regulatory submissions. The U.S. Food and Drug Administration provides guidance on the use of RWE for post-marketing surveillance, label expansion, and therapeutic approvals, while the European Medicines Agency promotes adaptive approaches for integrating RWE into regulatory decision-making processes. These initiatives reflect an increasing acknowledgment that traditional clinical trial settings may not fully represent real-world treatment effectiveness and safety profiles.
Despite these advancements, several challenges remain in the broader regulatory adoption of RWE. Real-world data must adhere to stringent standards related to data quality, statistical validity, traceability, and reproducibility. Consequently, many pharmaceutical companies continue to position RWE as a supplementary resource rather than a primary strategic component. To fully realize its potential across the drug development lifecycle, scalable, domain-specific, and regulatory-compliant AI-driven RWE solutions are becoming increasingly critical. Forward-looking organizations are actively leveraging these capabilities to transform the collection, analysis, and application of real-world data across the life sciences sector.
Read Comprehensive Report Overview: https://www.insightaceanalytic.com/report/ai-powered-real-world-evidence-rwe-solutions-market/3135
List of Prominent Players in the AI-Powered Real-World Evidence (RWE) Solutions Market:
- Aetion
- Atropos Health
- ConcertAI
- Envision Pharma Group
- Flatiron Health
- Health Compiler
- Huma
- IQVIA
- ai
- Komodo Health
- NVIDIA CLARA
- OM1
- ai
- Optum
- Owkin
- Panalgo
- Realyze Intelligence
- Syntropy
- Tempus
- AI
- Veradigm
- Other Prominent Players
Market Dynamics
Drivers:
The global healthcare industry is undergoing a transition toward value-based care models that prioritize clinical outcomes, quality of care, and cost efficiency over service volume. This paradigm emphasizes the use of evidence-based metrics to assess the effectiveness, efficiency, and overall value of healthcare interventions. Value-based frameworks are designed to improve patient outcomes while ensuring compliance with standardized performance benchmarks. In response to the limitations of traditional care delivery systems, healthcare providers are increasingly implementing strategies focused on enhancing patient outcomes and optimizing operational efficiency, thereby supporting the adoption of real-world evidence (RWE) solutions.
Challenges:
The expansion of the RWE market is constrained by a shortage of skilled professionals with expertise in advanced digital technologies and the analysis of complex healthcare datasets. The clinical research sector continues to face a talent gap in data analytics and interpretation, which limits the effective deployment and utilization of advanced RWE platforms in clinical trials and evidence-based decision-making processes.
Regional Trends:
North America is expected to maintain a leading position in the RWE market over the forecast period, supported by strong adoption of real-world data (RWD) among payers, well-defined regulatory frameworks, and a mature ecosystem of RWE solution providers. The region benefits from widespread application of RWE in drug development and regulatory submissions, a high prevalence of chronic diseases, advanced healthcare infrastructure, rigorous drug approval standards, and extensive use of electronic health records. Additionally, government initiatives aimed at promoting the integration of RWE continue to strengthen market growth.
The Asia-Pacific region is anticipated to register the highest growth rate during the forecast period. This expansion is driven by the presence of numerous contract research and manufacturing organizations in key markets such as China and India, along with increasing governmental support for the adoption of RWE. Growing demand for improved healthcare delivery, combined with strategic initiatives by industry participants—including regional partnerships, collaborations, and portfolio expansion—is further accelerating market development across the region.
Add our site to Google Preferred Sources for quality content: https://google.com/preferences/source?q=insightaceanalytic.com
Recent Developments:
- In Jan 2025, Atropos Health and xCures declared a collaboration to use RWE-based analytics to improve patient care. Through this collaboration, the businesses will use AI, the xCures Platform, and the Atropos EvidenceTM Network to create and distribute highly proven prognostic and decision support tools to healthcare providers throughout the healthcare ecosystem. The collaboration marks a major advancement in the analysis and use of RWE using AI. Atropos Health and xCures will collaborate to create solutions that provide traceable, source-verified insights straight into clinical processes from raw clinical notes, analytics, and imaging data.
- In July 2021, Syneos Health and Aetion declared a collaboration to offer analytics-driven solutions and regulatory-grade data to enhance medication development and improve patient outcomes. The collaboration combines Aetion Evidence Platform (AEP) with Syneos Health’s in-house data collecting and research tools to produce evidence through real-world evidence (RWE) analytics and patient data curation. Biopharmaceutical companies can reduce risk through evidence-based decision-making, speed up product development and patient access to new therapies, and prove value to a wide range of healthcare stakeholders, including patients, regulators, payers, and healthcare providers, with Syneos Health’s offerings, such as its Biopharmaceutical Acceleration Model, Dynamic Assembly, and RWE Solutions.
Global AI-Powered Real-World Evidence (RWE) Solutions Market – By Component
- Software/Platforms
- Services
Global AI-Powered Real-World Evidence (RWE) Solutions Market – By Data Source
- EHR/EMR Data
- Claims Data
- Genomics
- Wearables/IoT
- Patient-Reported Data
- Imaging
Global AI-Powered Real-World Evidence (RWE) Solutions Market – By Therapeutic Area
- Oncology
- Neurology
- Cardiology
- Rare Diseases
- Immunology
Global AI-Powered Real-World Evidence (RWE) Solutions Market – By End-User
- Pharma/Biotech
- Payers/PBMs
- Hospitals/IDNs
- Regulators
- CROs
Global AI-Powered Real-World Evidence (RWE) Solutions Market – By Deployment Model
- Cloud-Based
- On-Premise
- Hybrid
Global AI-Powered Real-World Evidence (RWE) Solutions Market – By Technology
- NLP
- Computer Vision
- Federated Learning
- Graph ML
Global AI-Powered Real-World Evidence (RWE) Solutions Market – By Application
- Drug Development
- Regulatory Submissions
- Market Access
- Precision Medicine
Global AI-Powered Real-World Evidence (RWE) Solutions Market – By Region
North America-
- The US
- Canada
Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
Asia-Pacific-
- China
- Japan
- India
- South Korea
- Southeast Asia
- Rest of Asia Pacific
Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
Middle East & Africa-
- GCC Countries
- South Africa
- Rest of the Middle East and Africa
Customize this Study according to your Requirements @ https://www.insightaceanalytic.com/customisation/3135
About Us:
InsightAce Analytic is a market research and consulting firm that enables clients to make strategic decisions. Our qualitative and quantitative market intelligence solutions inform the need for market and competitive intelligence to expand businesses. We help clients gain competitive advantage by identifying untapped markets, exploring new and competing technologies, segmenting potential markets and repositioning products. expertise is in providing syndicated and custom market intelligence reports with an in-depth analysis with key market insights in a timely and cost-effective manner.
Contact us:
InsightAce Analytic Pvt. Ltd.
Visit: https://www.insightaceanalytic.com/
Tel : +1 607 400-7072
Asia: +91 79 72967118
info@insightaceanalytic.com